 Air, water and steam are the most commonly used ways to distribute heat to process loads. However, steam offers several advantages hot air and hot water don’t have:
Air, water and steam are the most commonly used ways to distribute heat to process loads. However, steam offers several advantages hot air and hot water don’t have:
- The heat carrying capacity of steam is much greater than air or water
- Steam provides its own locomotive force
- Steam provides heat at a constant temperature
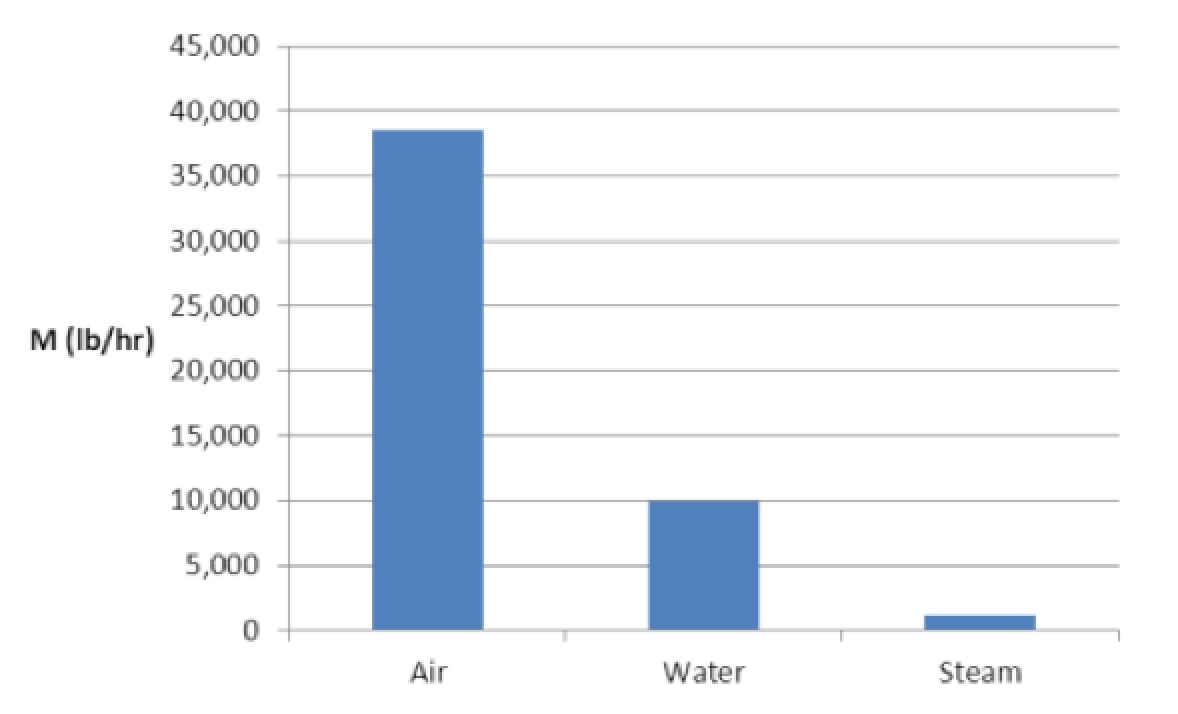
Msteam = Q / hfg = 1,000,000 Btu/hr / 881 Btu/lb = 1,135 lb/hr
HOT WATER: If the temperature of hot water dropped by 100º F as it passed through a heat exchanger, the mass flow rate of water to transfer the same amount of heat would be about nine times as much as steam:Mwater = Q / (cp x dt) = 1,000,000 Btu/hr / (1 Btu/lb-F x 100 F) = 10,000 lb/hr
HOT AIR: If the temperature of hot air dropped by 100º F as it passed through a heat exchanger, the mass flow rate of air to transfer the same amount of heat with the same temperature difference would be about 34 times as much as steam:Mair = Q / (cp x dt) = 1,000,000 Btu/hr / (0.26 Btu/lb-F x 100 F) = 38,500 lb/hr
 FLOW RATES: The higher flow rates required by water and air require pipes and ducts with larger diameters than steam pipes, which increases first cost and heat loss. In addition, air and water do not propel themselves. Thus, hot air and water distribution systems require fans or pumps, whereas a steam distribution system does not require any additional propulsion for outgoing steam and a very small pumping system for returning the condensate to the boiler.
CONSTANT TEMPERATURE: Finally, because steam condenses at a constant temperature, 100-psig steam could heat a process stream to a maximum temperature of 338º F which is the temperature of the steam. On the other hand, the temperature of water and air decrease as heat is transferred; thus, if the heat in these examples was delivered by a cross-flow heat exchanger, the maximum temperature of the process stream would be 100º F less than the incoming temperature of the air or water.
Because of these advantages, steam is the most widely used heat-carrying medium in the world.
FLOW RATES: The higher flow rates required by water and air require pipes and ducts with larger diameters than steam pipes, which increases first cost and heat loss. In addition, air and water do not propel themselves. Thus, hot air and water distribution systems require fans or pumps, whereas a steam distribution system does not require any additional propulsion for outgoing steam and a very small pumping system for returning the condensate to the boiler.
CONSTANT TEMPERATURE: Finally, because steam condenses at a constant temperature, 100-psig steam could heat a process stream to a maximum temperature of 338º F which is the temperature of the steam. On the other hand, the temperature of water and air decrease as heat is transferred; thus, if the heat in these examples was delivered by a cross-flow heat exchanger, the maximum temperature of the process stream would be 100º F less than the incoming temperature of the air or water.
Because of these advantages, steam is the most widely used heat-carrying medium in the world.
Why Steam is the Most Widely Used Heat-Carrying Medium in the World
